Bimzelx (Bimekizumab) is a monoclonal antibody, which calms down inflammation in psoriasis and psoriatic arthritis by binding and blocking two pro-inflammatory molecules:
- Interleukin-17A (IL-17A)
- Interleukin-17F (IL-17F)

Filling two needs with one dead, scientists developed an antibody against a site common for both A and F subtypes of interleukin 17. It allowed bimekizumab to completely shoutdown this signalling molecule from the inflammatory cascade.
What role Immune System Plays in Psoriasis ?

When our immune system attacks our tissues and organs, we call it an autoimmune disease.
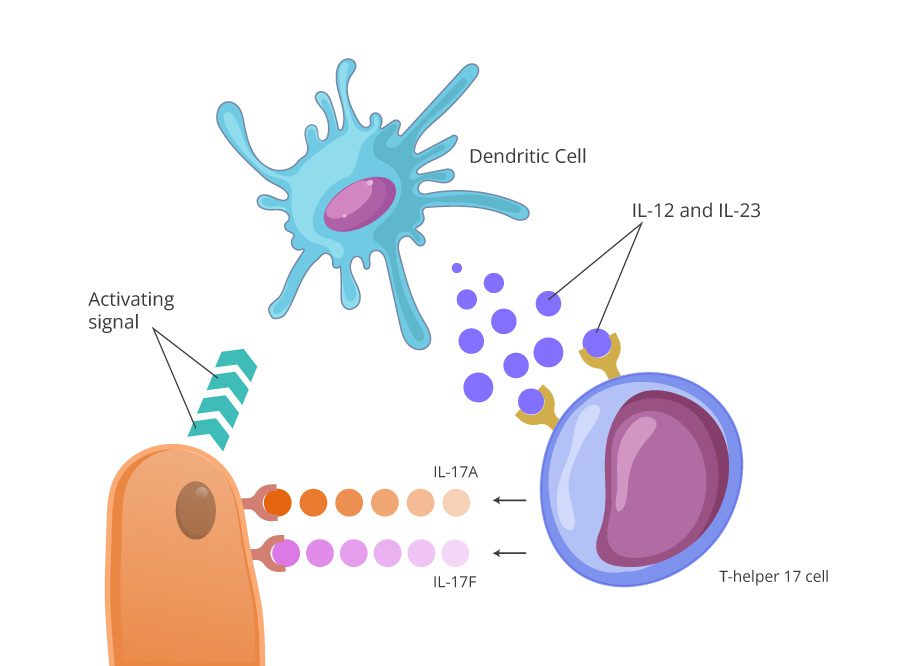
In psoriasis, the aggression of the immune system begins with the activation of dendritic cells in the skin. Those cells prevent infections from entering the body by engulfing them and presenting their antigens to plasma cells, which then produce bug-specific antibodies.
Dendritic cells
Being a first line of defense, they are tightly packed with different signalling molucules, that alert our defence system:
- TNF-α – tumor necrosing factor alpha
- Interleukin 12
- Interleukin 23

T-helper cells and IL-17
Interleukins 12 and 23 arouse another important cell in the IL-17 inflammation cascade – T-helper 17 cells (Th17). TH17 cells release six different types of IL-17, which scientists also refer as the IL-17 superfamily.
The main patriarchs of this family are IL-17A and IL-17F. They have a special landing place on the surface of the keratinocytes, called IL17C receptor (IL17R).
Keratinocytes
Besides being a main barrier of our body, kerytonocytes play important role in the immune response. If you cut your skin, you’re guaranteed to damage them. So keratynocytes should have a way to alert immune system about potential breach of skin integrity. They release several important components:
- antimicrobial peptides to fight infections localy
- cytokines, and chemokines, that act to reinforce and enhance immune response

Similar response happen when IL17A and IL17F land on the keratinocyte’s surface. They trigger release of chemicals activating even more dendritic cells. A vicious inflammatory cycle between the immune system and the skin is established, leading to several hallmark changes in psoriatic skin:
- Inflammation;
- Inflitration of the skin by immune cells;
- Keratinocyte proliferation – an accelarated skin cell turnover.

How Does Bimzelx Work
Bimekizumab is a humanized monoclonal IgG1 antibody that selectively inhibits interleukin (IL)-17A and IL-17F. Scientists have engineered the fragment antigen-binding (Fab) region to bind interleukins with high affinity. A link antibody + IL-17 squares IL-17 function away.

So Bimekizumab puts a break on a chain reaction of keratynocyte arousal, leading to postive clinical outcomes:
- Calming down skin iflammation. The result is a notable decrease in redness, swelling, and discomfort associated with inflammatory skin conditions.
- Halting abnormal prolifiration of Keratinocytes. Under inflammatory conditions, keratinocytes can multiply excessively, leading to the thick, scaly patches seen in psoriasis. By addressing IL-17 pathways, Bimzelx helps normalize keratinocyte turnover.
- Turn down autoimmune reponse. Bimzelx reduces this misguided immune response, which chill the psoriasis flare-ups down.
Approval of FDA and EMA
Bimekizumab has demonstrated superiority in all direct comparative clinical trials:
- Superior to ustekinumab (IL-12/23 inhibitor);
- Better than adalimumab (TNF inhibitor);
- Superior to secukinumab (IL-17A inhibitor).
Bimekizumab has received approval from both the FDA and EMA for its use in treating specific inflammatory conditions:
- Psoriasis
- Psoriatic arthritis
- Ankylosing Spondylitis
Clinical trials and outcome
In BE OPTIMAL, a multicenter, randomized, double-blind, placebo-controlled trial, Bimekizumab was to placebo in reducing joint, skin, and radiographic signs in patients with psoriatic arthritis who were naive to biologic treatments.
In one pivotal Phase 3 trial for plaque psoriasis, Bimekizumab demonstrated a higher rate of skin clearance compared to placebo groups. The primary endpoint, measured by the Psoriasis Area and Severity Index (PASI), highlighted that up to 90% of patients achieved at least a 75% reduction in PASI scores. These successful outcomes contributed to its subsequent approval.
Ankylosing Spondylitis Benefits
For ankylosing spondylitis, Bimekizumab offers notable benefits. Clinical studies have reported improvements in both objective and subjective measures of disease activity. Patients experienced significant reductions in the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) scores.
The drug works effectively to alleviate pain and stiffness associated with active ankylosing spondylitis. In trials, approximately 60-70% of patients reported achieving minimal disease activity after treatment. The sustained responses observed during follow-up periods further underscore its potential as a favorable treatment option for managing this condition.
Bimzelx Side Effects
Bimzelx (bimekizumab) can lead to various side effects. Understanding these effects is essential for informed decision-making regarding its use.
Short-Term Side Effects
Short-term side effects may occur after administering Bimzelx. Common effects include:
- Injection site reactions: Redness, itching, or swelling may appear at the injection site.
- Headache: Some users report experiencing headaches shortly after treatment.
- Nausea: Gastrointestinal discomfort is another notable concern.
- Fatigue: Feelings of tiredness can also manifest in some individuals.
These side effects are typically mild to moderate in severity. They often resolve on their own within a few days. Monitoring your response to the medication is essential.
Long-Term Safety Considerations
Long-term safety is a critical aspect of Bimzelx treatment. While many individuals tolerate Bimzelx well, some potential long-term side effects include:
- Infections: As an immunomodulator, Bimzelx may increase susceptibility to infections.
- Allergic reactions: Some individuals may develop allergic symptoms, such as rash or swelling.
- Changes in laboratory results: Regular monitoring of blood counts and liver function may be necessary.
Discussing these potential outcomes with your healthcare provider is vital. They can help you weigh the benefits against these risks based on your health profile.
Combinations with guselkumab and future of psoriasis treatment
Combining Bimekizumab with guselkumab represents a novel approach in psoriasis management. Both medications target different pathways in the immune response, making them suitable for combination therapy.
- Bimekizumab: Inhibits IL-17A and IL-17F.
- Guselkumab: Targets IL-23.
The interaction may enhance treatment efficacy, providing a more effective route for patients with moderate to severe psoriasis. Early studies suggest that dual inhibition can lead to faster and more significant skin clearance.
You may find that combination therapies can also help mitigate the risk of developing resistance to treatment over time. Personalized medicine remains a key focus, tailoring combinations based on individual patient responses.
Future studies will likely explore:
- Safety profiles: Understanding the long-term effects of combining these therapies.
- Efficacy outcomes: Assessing how combinations compare to monotherapies.
- Patient preferences: Gauging how patients respond to combined treatment regimens.
This ongoing research aims to optimize psoriasis management, providing you with more effective and customized treatment strategies. As science evolves, look out for emerging therapies that harness this combination approach.
Summary of Bimzelx Mechanism of Action
Bimekizumab is a monoclonal IgG1 antibody that selectively binds to interleukin (IL)-17A and IL-17F. By blocking IL-17 from activating keratinocytes, it calms down the inflammatory and leading to significant improvement of symproms in psoriasis and psoriatic arthritis.